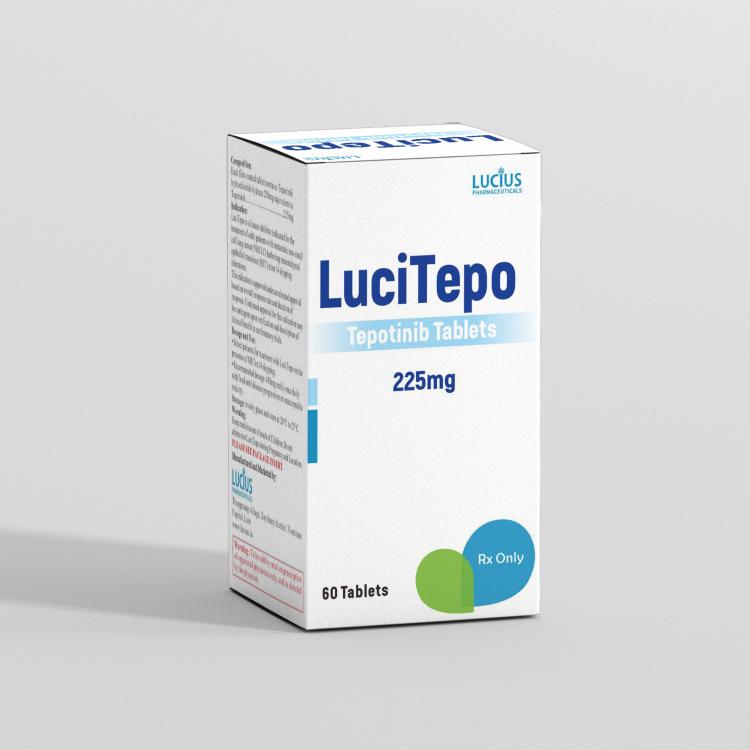
COMPOSITION:
Each film-coated tablet contains Tepotinib hydrochloride hydrate 250mg equivalent to Tepotinib…………………………….225mg.
INDICATION:
LuciTepo is a kinase inhibitor indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal epithelial transition (MET) exon 14 skipping alterations.
This indication is approved under accelerated approval based on overall response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
DOSAGE AND USE:
• Select patients for treatment with LuciTepo on the presence of METex14 skipping.
• Recommended dosage: 450mg orally once daily with food until disease progression or unacceptable toxicity.
STORAGE:
In a dry place and store at 20°C to 25°C.
MANUFACTURED AND MARKETED BY:
LUCIUS PHARMACEUTICALS(LAO) CO., LTD
No.26, Thongmang village, Xaythany district, Vientiane Capital, Laos
WARNING:
Keep medicine out of reach of Children.Do not administer LuciTepo during Pregnancy and Lactation.
Customer Reviews
Write a review
Cobus Bester
Cobus Bester on Mar 03, 2023
Can’t wait to start mixin’ with this one! Irba-irr-Up-up-up-up-date your theme!
Report as Inappropriate



.jpeg)

















.jpg)